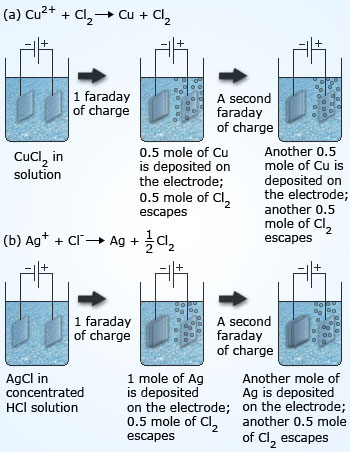
1 faraday charge is passed through aq solutions of AgNO3, Cuso, and Fecig. The ratio of g equivalents of Ag(s) : Cu(s): Fe(s) deposited is (1)/1: 1:1 (2) 6:3:2 (3) 1:2:3 (4) 1:2:1

Amazon.com: Cyber Nickel Copper 1 Faraday Fabric EMF Shielding 50" Width Signal Blocking Material - Plain Weave… (50" x 3')