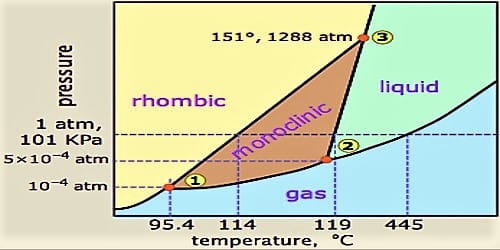
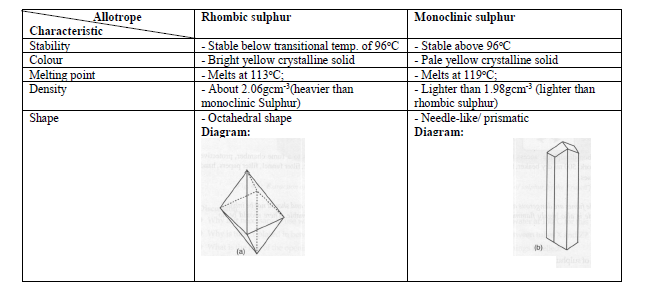
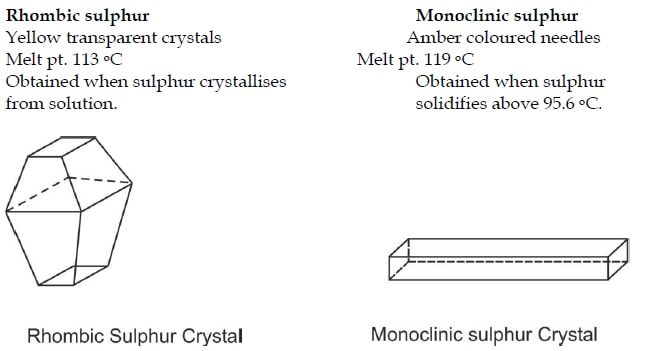
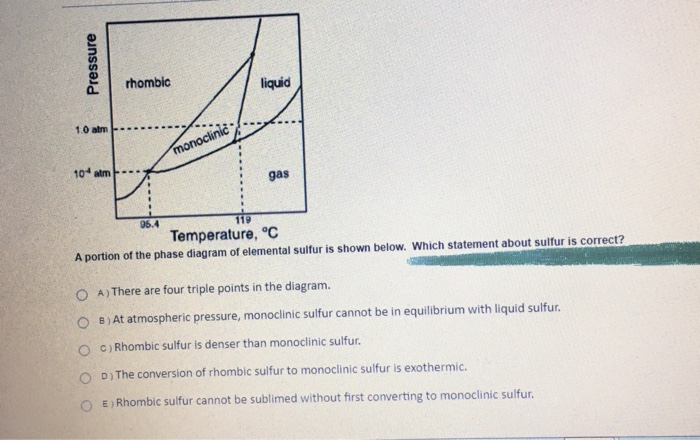
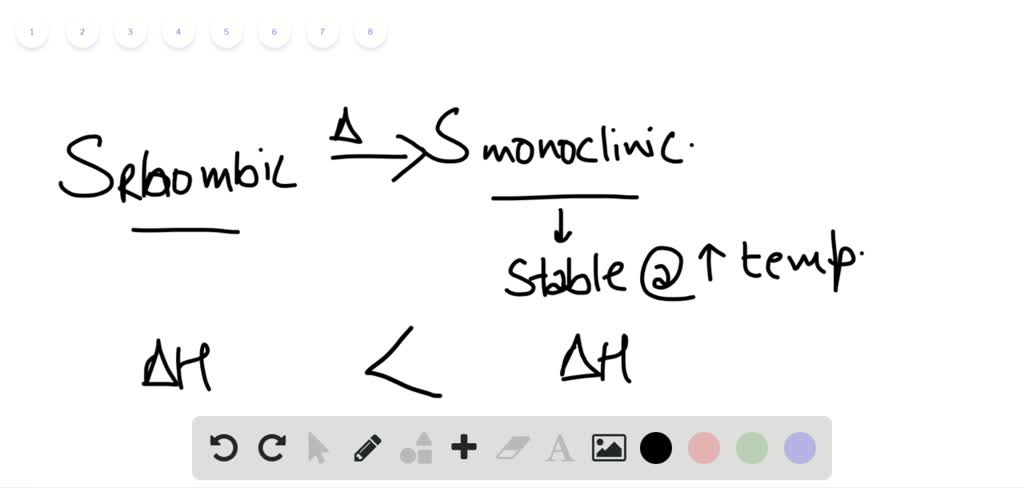
The heats evolved in combustion of rhombic and monoclinic sulphur are, respectively, 70960 and 71030cal mol^(-1). What will be the heat of conversion of rhomic sulphur to monoclinic?

39.The heats of combustion of rhombic and monoclinic sulphur are "70969" and "71040" calories respectively.What will be the heat of conversion of rhombic sulphur to monoodinic? 1) "7096" calories "," 2) "70130"

The heats of combustion of rhombic and monoclinic sulphur are - 70960 and - 71030 calorie respectively. What will be the heat of conversion of rhombic sulphur to monoclinic sulphur?

At 298 K the entropy of rhombic sulphur 32.04 J/mol K and that of monoclinic sulphur is 32.68 J/mol K. The heat of their combustion are respectively - 298246 and - 297948